|
2/28/2024 0 Comments Chromium iii hydroxidePlease solve b, and include the states (aq, l, g, s) as well as the correct arrow symbol (one way or both ways) as they are necessary for the answer. Chromium (III) Hydroxide is an inorganic compound with a green color and various applications in pigment, textile, and catalyst industries. The formula of chromium(III) hydroxide is Cr(OH)3. (Hint): Amphiprotic substances can behave as either an acid or a base. 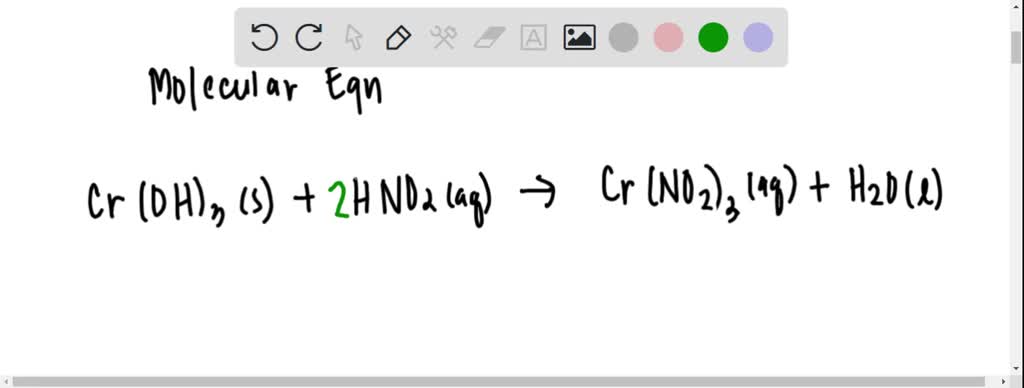
RIGHT ANSWER FOR a: Cr(OH)3(s) + 3H+(aq) → Cr+3(aq) + 3H2O(l)ī) Write a balanced chemical equation showing how an aqueous suspension of this compound reacts to the addition of a strong base. The formula of chromium(III) hydroxide is Cr(OH)3(s). chromium (III) hydroxide chromium (III) hydroxide Formula: CrH 3 O 3 Molecular weight: 103.0181 CAS Registry Number: Information on this page: Notes Data at other public NIST sites: X-ray Photoelectron Spectroscopy Database, version 4. If instead a strong acid is added to the solution, the added H + will react essentially completely with A to form HA. 
(III) was the dominant species in these systems however, this may not always. In this video we'll write the correct formula for Chromium (III) hydroxide : Cr(OH)3.To write the formula for Chromium (III) hydroxide we’ll use the Periodic. Because of the reaction described in Equation 18.7.1 18.7.1, the predicted solubility of a sparingly soluble salt that has a basic anion such as S 2, PO 43, or CO 32 is increased. (Hint): Amphiprotic substances can behave as either an acid or a base. Chromium(VI), chromate, is highly toxic to humans and is quite soluble and. The kinetics of the anation of the chromium(III) species, Cr(H 2 O) 63+ and Cr(H 2 O) 5 OH 2+, by hexacyanoferrate(II) ions in the pH range 4.55.4 are. A) Write a balanced chemical equation showing how an aqueous suspension of this compound reacts to the addition of a strong acid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
